
Homemade explosives (HMEs) are attractive to criminals and terrorists due to accessible online recipes, readily available equipment, and the ease of obtaining precursor materials. To counter this, EU Regulation 2019/1148 restricts access to high concentrations of “Restricted Precursors” and monitors sales of “Reportable Explosives Precursors”.
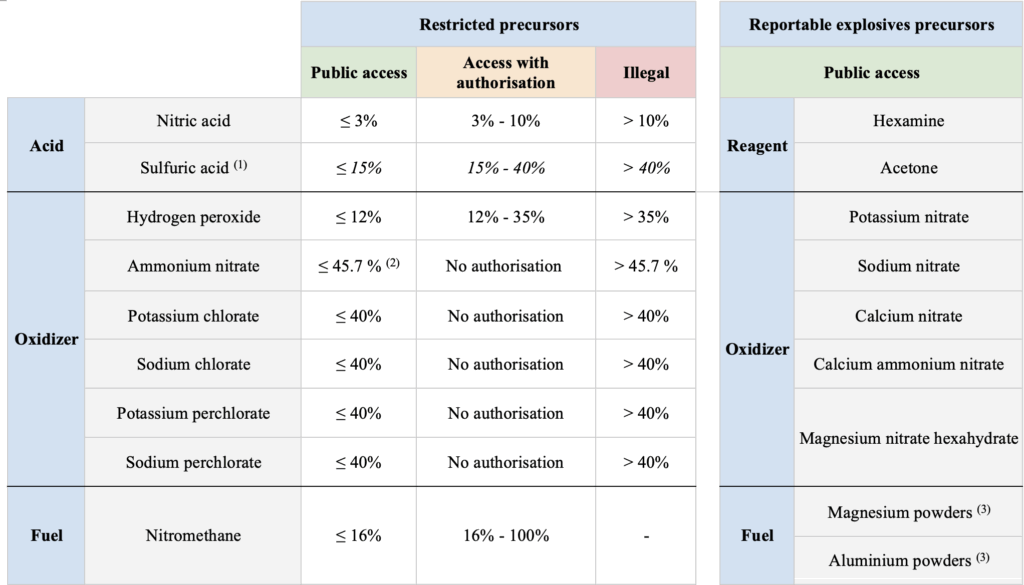
- (1) Sulfuric acid is not restricted in Switzerland (RS 941.421, 2022).
- (2) 16% w/w nitrogen from ammonium nitrate corresponds to 45.7% ammonium nitrate, after removal of impurities.
- (3) With a particle size of less than 200 μm, and for a substance or in mixtures containing 70% w/w or more of aluminium or magnesium.
Classification as an explosive precursor depends on concentration, making a product legal or illegal based on thresholds where the risk of illicit explosive manufacturing outweighs legitimate public use. The article, published in Forensic Science International (2025), emphasizes the added value of on-site detection and quantification: it enables first responders to quickly and safely identify substances and determine if they exceed legal limits, assessing legality and potential hazards. This is crucial for taking appropriate safety measures, mitigating risks, and disrupting HME production, especially given challenges with traditional portable devices like Raman (e.g., high costs, fluorescence interference, and safety risks with energetic materials). A similar approach has already proven highly effective in on-site drug detection using portable NIR spectroscopy, as detailed in our post Revolutionizing Forensic Analysis with Ultra-Portable NIR Technology.
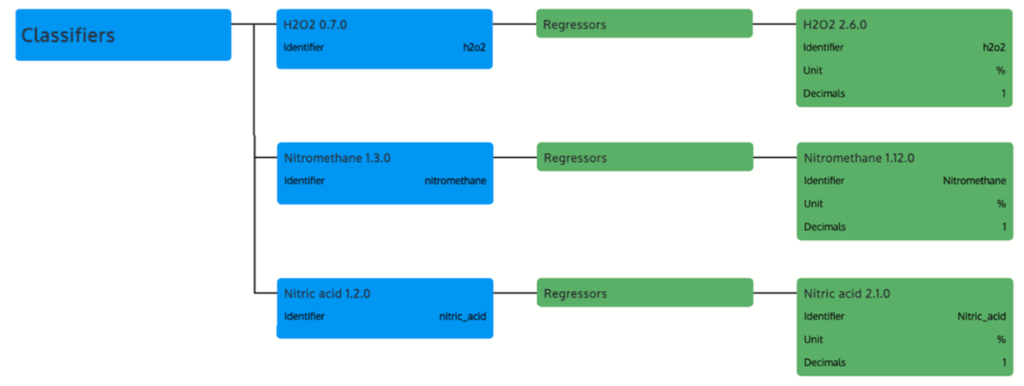
The study evaluates portable near-infrared (NIR) spectroscopy combined with machine learning for detecting and quantifying hydrogen peroxide (H2O2), nitromethane (CH3NO2), and nitric acid (HNO3), achieving high accuracy in diverse samples.
Methodology Overview
The article utilized the MicroNIR OnSite-W device (950–1650 nm range) with a droplet accessory for 100 μL liquid samples, analyzing each three times across devices to account for variability. Samples encompassed laboratory-grade dilutions and commercial products: H2O2 (1–60% v/v, including hair oxidants and stain removers), CH3NO2 (0.5–99% v/v in methanol, including model fuels), and HNO3 (3–65% v/v in water). Reference methods included titration for H2O2and GC-MS for commercial CH3NO2.
A sequential approach involved classification models for identification, followed by substance-specific regression for quantification, tested against non-target sets for selectivity.
Key Findings: Classification and Regression Results
Classification (Qualitative Models)
The study implemented stacking models for binary classification (analyte vs. non-analyte) to ensure accurate identification before quantification, using datasets of 67 H2O2 samples (44 original + 23 additional), 36 CH3NO2, and 12 HNO3, plus large non-target sets (n=1009–1068, including water, acids, other precursors, and non-explosives).
- H2O2: Achieved overall accuracy of 0.994 on calibration and validation sets (Table 2). No false positives; minimal false negatives (low-concentration samples, non-critical as they are legal and low-risk). High precision (1.000), recall (0.989), and F1-score (0.994) demonstrate reliability and selectivity.
- CH3NO2: Overall accuracy of 0.998 (Table 3). One false positive (methanol misclassified as CH3NO2) and minimal false negatives, with precision (0.998), recall (0.998), and F1-score (0.998) indicating excellent performance across lab and commercial samples.
- HNO3: Overall accuracy of 0.997 (Table 4). No false positives; minimal false negatives (low concentrations), with precision (1.000), recall (0.993), and F1-score (0.997).
These results confirm the models’ robustness, with low error rates enabling confident on-site identification aligned with legal thresholds.
Regression (Quantitative Models)
Following classification, regression models quantified concentrations, yielding low errors and high correlations:
- H2O2 model produced RMSEP=0.96%, r²=0.99 (Figure 5). Limit of Detection (LOD)=2.57%, Limit of Quantification (LOQ)=7.78%, suitable for distinguishing below/above the 12% legal threshold for public access.
- CH3NO2 model gave RMSEP=2.46%, r²=0.99 (Figure 7). LOD=5.76%, LOQ=17.45%, effective for the 16% threshold, handling additives in commercial fuels.
- HNO3 model resulted in RMSEP=0.70%, r²=0.99 (Figure 9). LOD=2.35%, LOQ=7.12%, precise for the 3% threshold.
Predictions closely matched references across ranges, with models adaptable to variability via cloud updates.
Implications for Forensic Analysis
The article highlights how this NIR-machine learning approach provides rapid, non-destructive on-site tools for assessing legality and hazards, integrating with cloud systems for real-time data sharing and forensic intelligence. It addresses traditional method limitations while supporting regulatory compliance.
The study suggests extending models to more precursors, higher concentrations, and complementary techniques like Raman to enhance applicability and safety.
Conclusion: Advancing Forensic Response with Portable NIR Technology
This study demonstrates how portable NIR spectroscopy combined with machine learning can significantly enhance first responder capabilities in identifying and quantifying explosive precursors on-site. By enabling rapid, non-destructive, and legally relevant assessments, these tools support safer decision-making, faster interventions, and more efficient use of laboratory resources.
As highlighted in Forensic Science International, this approach marks an important step toward modernizing forensic workflows and strengthening frontline detection efforts. NIRLAB provides robust, field-ready solutions that empower agencies to detect hazardous materials with accuracy and confidence.
To learn more about how NIRLAB’s technology can support your forensic or law-enforcement operations, we invite you to explore our other articles or contact us directly at contact@nirlab.com for tailored guidance


