Introduction
Near-infrared (NIR) spectroscopy is a powerful analytical technique that has found wide applications in various fields, including chemistry, pharmaceuticals, agriculture, food science, and materials science. It involves the interaction of near-infrared light with matter to obtain valuable information about its molecular composition and structure.
NIR spectroscopy utilizes the portion of the electromagnetic spectrum ranging from approximately 780 to 2500 nanometers (nm), which lies adjacent to the visible light region. This region is characterized by low-energy photons, and the corresponding wavelengths are longer than those used in visible or ultraviolet spectroscopy. The interaction of NIR light with a sample can be primarily attributed to overtones and combinations of molecular vibrations.
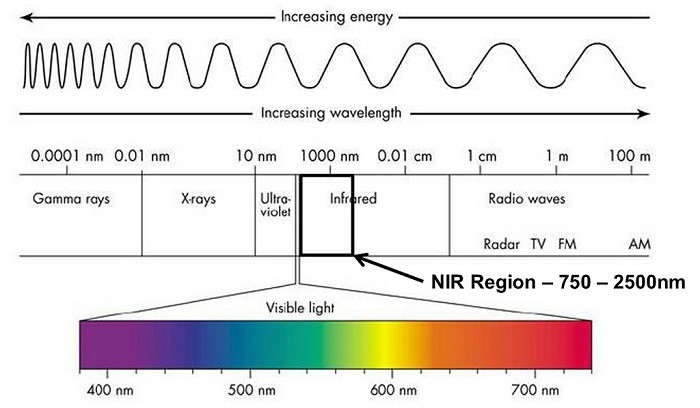
Elecromagnetic spectrum with NIR band highlighted
The fundamental principle behind NIR spectroscopy is based on the fact that different chemical compounds absorb and scatter light in the NIR region in a characteristic manner. This interaction is influenced by the presence of specific molecular bonds, functional groups, and molecular arrangements within the sample. By analyzing the absorption and scattering patterns of NIR light, it is possible to identify and quantify various chemical constituents in a sample.
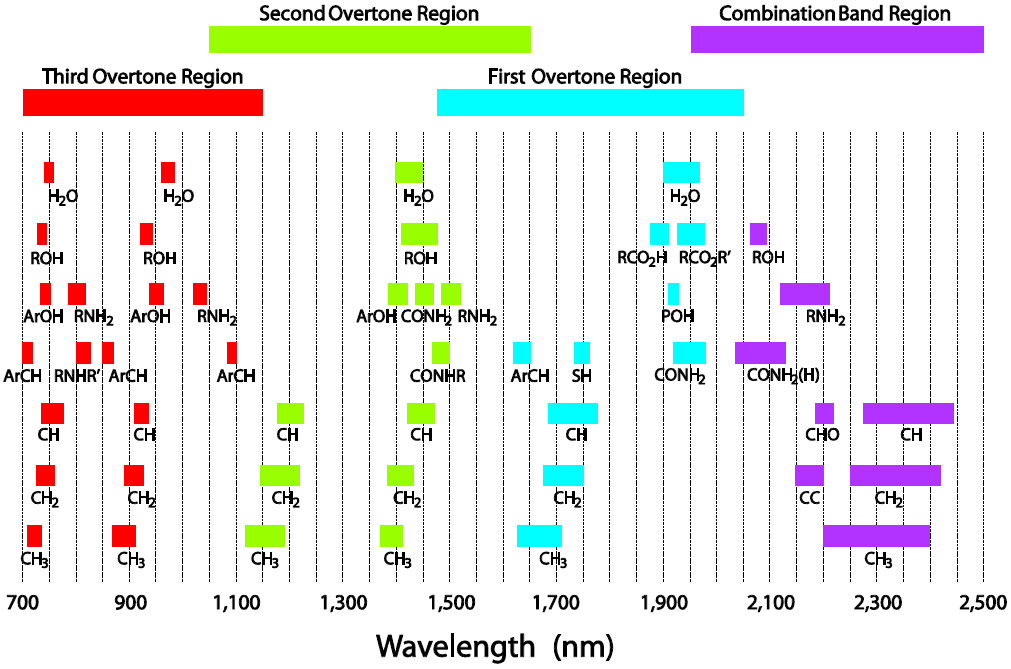
Regions of NIR in respect of the functional groups involved
One of the major advantages of NIR spectroscopy is its non-destructive nature, as it requires minimal or no sample preparation. This makes it highly suitable for rapid analysis of a wide range of samples, including liquids, solids, and gases. Additionally, NIR spectroscopy is applicable to both organic and inorganic compounds, enabling its use in diverse areas.
The instrumentation for NIR spectroscopy typically consists of a light source, which emits NIR radiation, a sample holder or cuvette, and a detector that measures the intensity of the transmitted or reflected light. The collected data is then processed using advanced mathematical algorithms and compared to a reference database or calibration model to obtain accurate qualitative and quantitative information.
Precise and rapid determination requires high-performance mobile NIR solutions such as FIELDLAB by NIRLAB.
Applications
NIR spectroscopy finds numerous applications in various industries. In pharmaceuticals, it is used for drug quality control, analysis of raw materials, formulation development, and monitoring of manufacturing processes. In agriculture and food science, NIR spectroscopy is employed for assessing the nutritional content, moisture, and chemical composition of crops, as well as for evaluating the quality of food products. It is also utilized in environmental analysis, forensics, and material characterization, among other fields.
In conclusion, NIR spectroscopy is a versatile and non-destructive analytical technique that provides valuable insights into the chemical composition and structure of a wide range of materials. Its speed, ease of use, and broad applicability make it an indispensable tool in many scientific and industrial settings.


